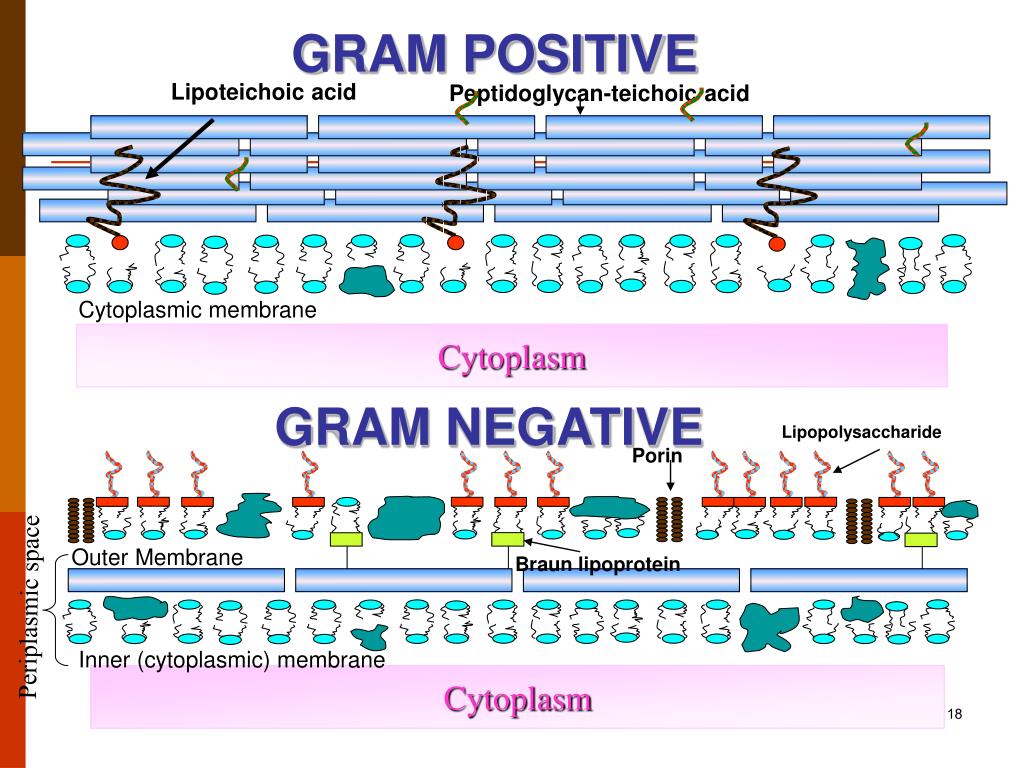
LTA and LPS are both anchored via their glycolipids to the membrane and carry a polysaccharide chain extending into the PGN layer of the cell wall. LTA is particularly interesting as a Gram-positive “endotoxin” because it resembles LPS in some respects. Indeed, LTA and PGN from Staphylococcus aureus have been demonstrated to interact with TLR2 and the TLR coreceptor CD14 ( 8, 9). Because PGN and LTA, teichoic acid or similar anionic molecules are shared by virtually all Gram-positive bacteria, these molecules are candidates as conserved toxins or “pathogen-associated molecular patterns” that are widely believed to be recognized by TLRs ( 7). 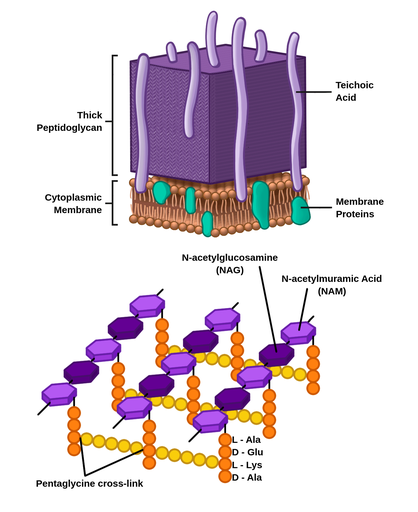
The cell wall of Gram-positive bacteria constitutes a macromolecular assembly of surface proteins, cross-linked peptidoglycan (PGN), polyanionic teichoic acid, and lipoteichoic acid (LTA) ( 6). In conclusion, LTA from GBS is a TLR2/TLR6 ligand that might contribute to secreted GBS activity, but does not contribute significantly to GBS cell wall mediated macrophage activation. 
Furthermore, GBS expressing LTA devoid of d-alanine residues, that are essential within immune activation by LTA, exhibited similar inflammatory potency as GBS with alanylated LTA. aureus, specifically differences in glycosyl linkage, in the glycolipid anchor and a lack of N-acetylglucosamine substituents of the glycerophosphate backbone. Structural analysis by nuclear magnetic resonance spectrometry revealed important differences between LTA from GBS and S. However, LTA from GBS proved to be a relatively weak stimulus of phagocytes containing ∼20% of the activity observed with LTA from Staphylococcus aureus. Furthermore, NF-κB activation by LTA required the integrity of two putative PI3K binding domains within TLR2 and was inhibited by wortmannin, indicating an essential role for PI3K in cellular activation by LTA. Using cells from knockout mice and genetic complementation in epithelial cells we found that highly pure LTA from both GBS and Staphylococcus aureus interact with TLR2 and TLR6, but not TLR1, in contrast to previous reports. In this study we examined the role of LTA from GBS in phagocyte activation and the requirements for TLR-LTA interaction. In contrast, the cell wall component lipoteichoic acid (LTA) from diverse Gram-positive bacterial species has been shown to engage TLR2. Group B Streptococcus (GBS) cell walls potently activate phagocytes by a largely TLR2-independent mechanism.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
